
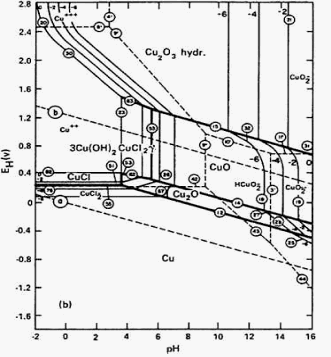
Various fillers including nanofillers reinforce the pristine polymer keeping in contact with its original properties. Filled systems circumvent these shortcomings with improved properties without additional cost. Often, there is a trade-off between the requirement and actual properties of pure materials. Polymeric materials, being light weight, are replacing the conventional metal and ceramics, especially in an aerospace application. The diagrams were compared with the simple Cu-H2O system at 25☌.The requirement of materials for aerospace industries includes light weight, high reliability, durability against corrosion, vacuum, radiation, fatigue and passenger safety. Pourbaix diagrams (electrode potential-pH diagrams) for Cu-Br?-H2O systems at 25☌ were developed in 400-g/L and 700-g/L (4.61-M and 8.06-M) LiBr solutions, common concentrations in different parts of refrigeration plants. The goal of the present work was the prediction of the general conditions of immunity, passivation, and corrosion for copper in aqueous LiBr-concentrated solutions. Thermodynamic aspects of corrosion can be described by electrode potential-pH diagrams, known as Pourbaix diagrams, which are graphical representations showing regions of thermodynamic stability of species in metal-water electrolyte systems.4 These diagrams can be constructed by calculations based on the Nernst equation and on the equilibrium constant of diverse metallic compounds. Lithium bromide (LiBr) is one of the absorbents most widely used in refrigeration technology.1 Although LiBr possesses favorable thermophysical properties, LiBr solutions can cause serious corrosion problems on structural materials (copper, steels, and other metals) in such plants. The effect was enhanced at higher bromide ion activities. Comparison of the simple Cu-H2O system with the diagrams for Cu-Br?-H2O systems at 25☌ showed that the formation of CuBr2 complexes extended the copper solubility range to both higher pH values and lower potentials by destabilizing the formation of copper oxides and promoting more active behavior of the metal. Activities of all the ion species containing copper were plotted for 10?6, 10?4, 10?2, and 100. Equilibria for Cu-Br?-H2O systems at 25☌ were determined for bromide ion activities of 15.61 and 194.77, which correspond to 400-g/L and 700-g/L LiBr solutions, respectively. Conventional procedures were followed to calculate the electrochemical and chemical equilibria from standard Gibbs free energy data. Pourbaix diagrams were constructed from standard Gibbs free energy data ( ?G°) of all the species considered. The diagrams were compared with the simple Cu-H2O system at 25☌. Pourbaix diagrams (electrode potential-pH diagrams) for Cu-Br?-H2O systems at 25☌ were developed in 400-g/L and 700-g/L (4.61-M and 8.06-M) lithium bromide (LiBr) solutions, common concentrations in different parts of refrigeration plants.

 RSS Feed
RSS Feed
